 * Some caveats: for example, aluminium will react slowly with water, if the thin aluminium oxide layer that prevents it from reacting is damaged. 2.1.1 recall the reactivity series of metals, including K, Na, Ca, Mg, Al, Zn, Fe and Cu 2.1.3 explain how the reactivity of metals is related to the tendency of a metal to form its positive ion 2.1. * This graphic doesn’t contain every metal in the periodic table. Majority of metals occur naturally in compounds, which we must remove them from. Some metals are so unreactive they occur largely uncombined with other elements, simple to obtain. This is because they can react with the compounds in metal ores, and displace the metals, aiding with their extraction. Carbon and hydrogen are also shoehorned in between entries in the list, despite being non-metals. Copper sulfate + zinc → zinc sulfate + copper Magnesium sulfate + zinc → NO REACTION The reactivity series also gives us an insight into why different metals are extracted from their ores in different ways. 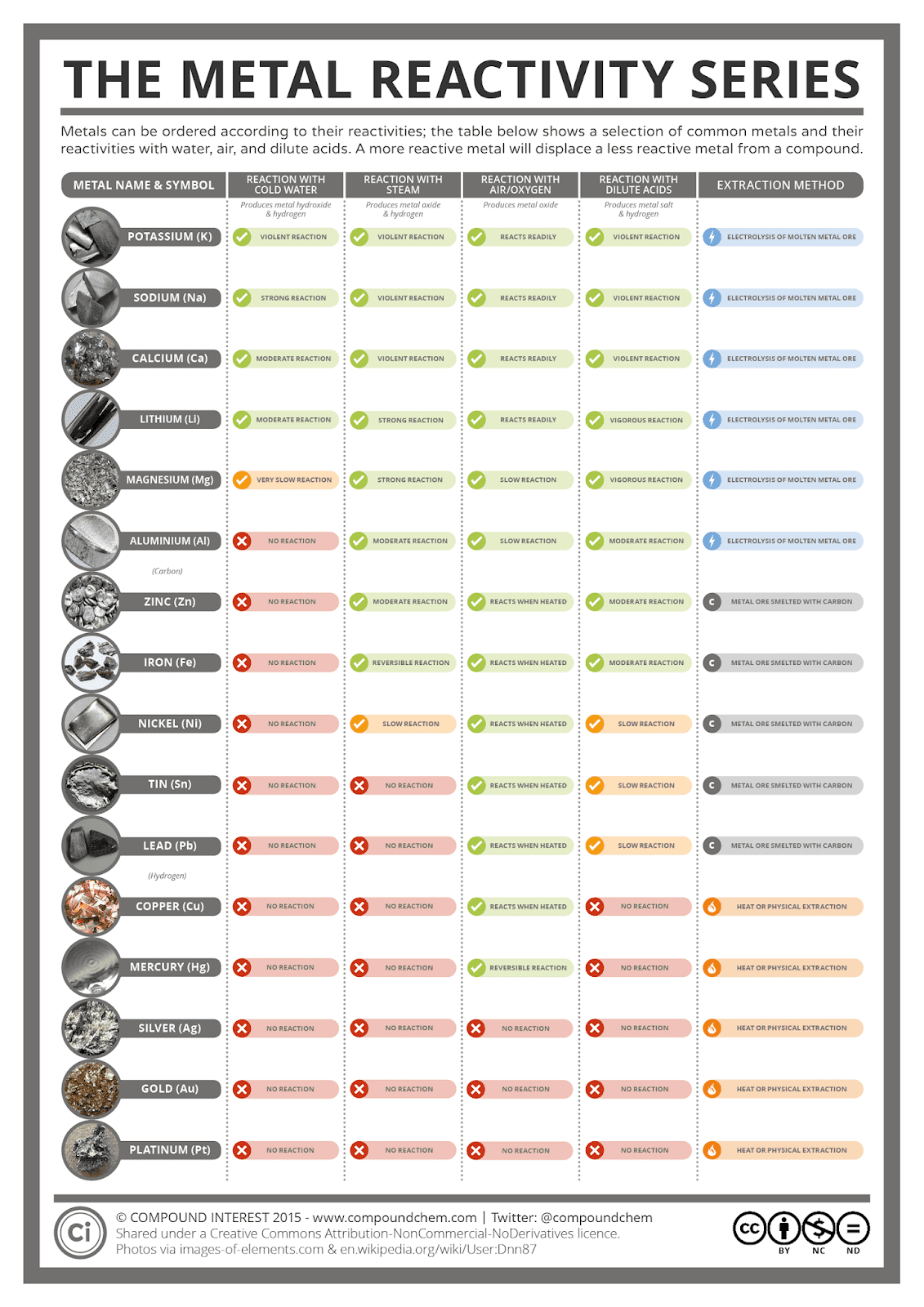
Conversely, if we react a metal with another metal lower in the series, no reaction will take place. The more reactive metal will take the place of the less reactive metal in the compound. If a metal compound reacts with a metal that’s above it in the reactivity series, a displacement reaction will occur. The table 7.11.1 below is an activity series of most common metals, and the table 7.11.2 is an activity series of the halogens. Since metals replace other metals, while nonmetals replace other nonmetals, they each have a separate activity series. Uses of this: * predict the outcome of certain chemical reactions. The activity series is a list of elements in decreasing order of their reactivity. Transition metals are much less reactive Gold and platinum have little in the way of chemical reaction. Closely followed by the marginally less reactive group 2 metals. Metals have a range of reactivities: Videos: the classic alkali metals in water demonstration The reactivity series offers a ranking of the metals in order of their reactivity. Other factors may, however, come into play as metal potassium can be prepared by reducing sodium potassium chloride to 850 ° C.Īlthough sodium in the reactivity series is less than potassium, the reaction can continue since potassium is volatile and the mixture is distilled.The text below has been excerpted from Compound Chemistry Student handout: Reactivity Series of Metals (PDF) This graphic places a selection of common metals into order of reactivity, as well as showing their reactions with air, water and steam. Likewise, the removal of titanium from tetrachloride can be achieved using magnesium, which in the end forms magnesium chloride: For instance, iron(III) oxide is decreased into iron and aluminium oxide is converted in this process. (Ti is approximately the same level as Al in the reactivity series). This is used for the thermite reaction for the production of small amounts of metallic iron and for the preparation of titanium by kroll process 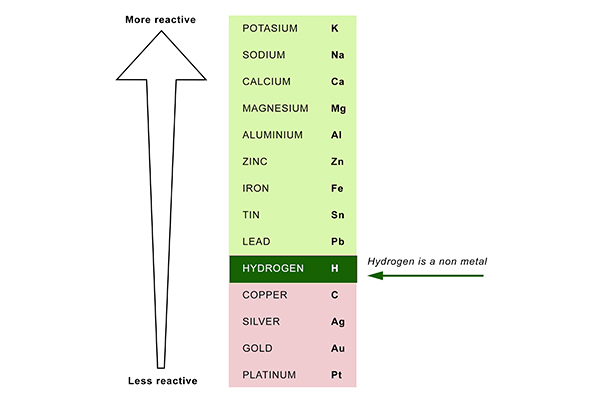
Generally, any of the metals that are lesser in the reactivity series can be replaced by metal: higher metals reduce lower metal ions. An iron nail in a copper sulphate solution can soon change colour as metallic copper is coated with the iron(II) sulphate. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
