Ionic bonds: bonds formed by the transfer of electrons. Valence Electrons: electrons in the outermost energy level of an atom. Nucleus: central core made of protons and neutrons. Proton: a particle with a positive charge.ħ Compounds Neutron: neutral particle, does not have a charge.Įlectron: has a negative charge. Atom: the smallest unit of an element that has all the properties of the element. 
Chemical Formula: uses chemical symbols and subscripts to identify the number of atoms of each element in a molecule of a compound. Compound: a type of matter that forms when two or more elements combine chemically.Ħ Compounds Molecule: the smallest unit of a compound that has all the properties of the compound. Mixture: a type of matter that forms when two or more substances are combined but do not join together chemically. Synthetic element: elements that are made by scientists in a laboratory and do not exist in nature. Element: a substance that cannot be broken down into simpler substances by ordinary chemical means. Matter: anything that has mass and volume. 
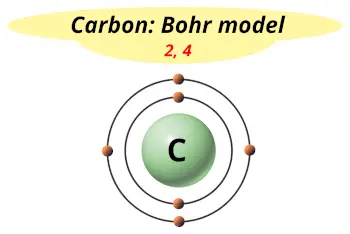
Synthetic Chemical: a chemical that is not formed in nature and is made by people. Example: Carbon C Hydrogen H Oxygen O Carbon C Iron Fe Sodium Na Magnesium MgĤ Identify Chemicals Chemical: substance used in or formed by a chemical process. Example: Carbon Cģ List common elements & chemical symbols.Ĭhemistry List common elements & chemical symbols. Example: Carbon CĬhemistry List common elements & chemical symbols. Presentation on theme: "Chemistry Draw a Bohr Model of a carbon atom."- Presentation transcript:ġ Chemistry Draw a Bohr Model of a carbon atom.Ģ List common elements & chemical symbols. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
